Studies have
repeatedly demonstrated that sensitization
to fungi, such as Alternaria, is
strongly associated with allergic rhinitis
and asthma in children (Arshad et al. 2001;
Downs et al. 2001; Halonen et al. 1997;
Nolles et al. 2001). However, the role of
exposure to fungi, measured in environmental
samples, in the development of childhood
allergic rhinitis is poorly understood.
Allergic rhinitis affects an estimated 20-40
million people in the United States alone,
and the incidence is increasing. Allergic
rhinitis has been associated with snoring,
sleep apnea, and sleep disturbances in
children (Camhi et al. 2000; Corbo et al.
2001; Scharf and Cohen 1998). The prevalence
of symptoms of allergic rhinoconjunctivitis
ranged between 15 and 25% among children
13-14 years of age who lived in the United
States or the United Kingdom (International
Study of Asthma and Allergies in Childhood
1998). A recent study documented that 93% of
adolescents with allergic asthma also have
allergic rhinitis. The diagnosis of allergic
rhinitis preceded asthma in up to 64% of
patients with both diseases (Kapsali et al.
1997). Because allergic rhinitis is a common
disease that lessens quality of life and is
associated with significant morbidity,
determining environmental exposures
associated with the development of allergic
rhinitis is of great public health
importance.
In a
prospective birth-cohort study of children with
parental history of asthma or allergies, we
assessed environmental fungal exposure
(culturable fungi in house air and dust samples)
and evaluated whether high fungal levels were
independently associated with doctor-diagnosed
allergic rhinitis in the first 5 years of life.
Study
protocol. Participants were part of a
metropolitan Boston, Massachusetts, prospective
birth-cohort study designed to examine
relationships between amounts of indoor
allergens, including fungi, and the development
of allergy and asthma. The study was approved by
the institutional review board of Brigham and
Women’s Hospital. With the consent of the
primary caregiver, screening and recruitment of
the families, which we have previously described
(Gold et al. 1999), was conducted between
September 1994 and June 1996. Monday through
Friday, all mothers who delivered at Brigham and
Women’s Hospital, a large Boston hospital, were
approached for screening within 24-48 hr after
delivery. Eligibility criteria included
residence inside route 128 (a highway encircling
the Boston metropolitan area); maternal age ≥ 18
years; history of hay fever, asthma, or
allergies in either parent; and maternal ability
to speak English or Spanish. Families were not
screened if the newborn was hospitalized in the
intensive care unit, was of gestational age < 36
weeks, or had a congenital anomaly. One month
after birth, a questionnaire regarding the
child’s health was administered by telephone to
families who had initially expressed interest in
participating in the study and who fit the
inclusion criteria. At 1 month, parents who
expressed plans to move or lack of interest in
the study were also excluded. Of the 1,405
families screened, 906 were excluded from the
study before the initial home visit. Reasons for
exclusion were reluctance to participate in a
longitudinal study (51%), plans to move within 1
year (39%), early loss to follow-up (9%), and
other (1%). After written informed consent was
obtained, a home visit was made when the child
was 2-3 months of age, and a questionnaire
regarding health outcomes, home characteristics,
environmental exposures, smoking, and
demographics was administered by trained
research assistants. Every 2 months, beginning
when the child was 2 months old and continuing
until the second birthday, a telephone
questionnaire was administered to the child’s
primary caretaker. After that, health outcomes
were ascertained semiannually.
At the initial
home visit, indoor air samples were collected
from each home using a Burkard culture plate
sampler (Burkard Manufacturing Co. Ltd.,
Hertfordshire, UK) operated at 45 L/min
[calculated cut point, diameter allowing 50% (D50)
= 2 µm] that collected particles onto DG18 agar
(a glucose medium containing dichloran and 18%
glycerol) in 90-mm Petri dishes. Sequential
duplicate 1-min air samples were collected in
the bedroom 1-1.5 m above the area of the floor
demarcated for dust collection. After sampling,
the Petri plates were returned to the laboratory
on the same day for incubation. The sampler
sieve plate was cleaned with an isopropanol swab
after each home visit.
After air
samples were collected, 2 m2 of the
floor surrounding the newborn’s bed was vacuumed
for 5 min, using a Eureka Mighty-Mite canister
vacuum cleaner (Eureka Co., Bloomington, IN)
modified to collect dust in a 19-
 90-mm cellulose
extraction thimble (Whatman International Ltd.,
Herefordshire, UK). In cases where both a smooth
floor and a rug were present, 2.5 min was
devoted to sampling the rug and 2.5 min was
spent vacuuming the smooth floor surface. After
sampling, the thimble was sealed in a plastic
bag and returned to the laboratory for sifting
(425-µm mesh sieve) and weighing.
90-mm cellulose
extraction thimble (Whatman International Ltd.,
Herefordshire, UK). In cases where both a smooth
floor and a rug were present, 2.5 min was
devoted to sampling the rug and 2.5 min was
spent vacuuming the smooth floor surface. After
sampling, the thimble was sealed in a plastic
bag and returned to the laboratory for sifting
(425-µm mesh sieve) and weighing.
A weighed
aliquot of fine dust from the dust sampling was
suspended (~ 25 mg/mL) and serially diluted
(e.g., full strength, 1:10, and 1:100) in an
aqueous solution of 0.02% Tween. Duplicates of
each dilution were spread-plated on DG18 culture
media (Verhoeff et al. 1994). After a mean ± SD
incubation period of 27.9 ± 14.5 days, we
identified fungal colonies on both air- and
dust-sample plates to genus (subgenus in the
case of Aspergillus isolates) using
standard mycologic criteria (Ellis 1971; von Arx
1970). We adjusted the number of colonies
recovered on the air sample plates for possible
multiple impactions using the positive-hole
correction equation (Andersen 1958). The
calculated concentrations of dust-borne fungi
were the number of colony-forming units (cfu)
per gram of dust, and those of airborne fungi
were cfu per cubic meter of air. We prioritized
the use of limited amounts of dust collected
from a home so that measurement of allergens for
cockroach, cat, and dust mites could be
performed first (Chew et al. 1998), followed by
analyses of fungi and endotoxin, respectively.
If there was not enough dust for allergen
analyses, then the fungal analyses were
performed first. Thus, when there was > 200 mg
or < 25 mg dust collected, fungal concentrations
were determined. There were 84 children
eliminated because the amount of dust collected
was between 25 and 200 mg.
Fungal
data. In previous analyses of these data
(Stark et al. 2003), we found that it was useful
to create indices indicating high levels of
fungi in the home. The variable was coded 1 if
that home produced “high” levels of that fungus,
and 0 otherwise. We define “high” as greater
than the 90th percentile for each taxon. Based
on Akaike’s Information Criterion (Hastie and
Tibshirani 1990) and -2 log likelihood, models
with indices for high fungal levels fit the data
better than do models treating the fungal
concentrations as continuous or natural
logarithm-transformed data.
Definition of allergic rhinitis and predictor
variables. We assessed allergic rhinitis
from 12 to 60 months of age by asking “Has your
child had allergic rhinitis or hay fever
diagnosed by a doctor?” Children without
12-month follow-up (n = 10) were
excluded, leaving 405 children with > 12 months
of health outcome data, as well as fungal data.
Definitions of other predictor variables.
Sociodemographic, familial, and perinatal
variables considered for inclusion in the
multivariate analysis of the relation between
indoor fungal exposure and allergic rhinitis
included sex, season of birth [defined as winter
(December-February), spring (March-May), summer
(June-August), or fall (September-November)],
race/ethnicity of the child according to
parental reporting (Litonjua et al. 1998), total
annual family income (< $30,000, $30,000-50,000,
and > $50,000), active maternal history of
asthma (physician-diagnosed asthma and wheeze in
the past year), maternal sensitization to
Alternaria [specific immunoglobulin E (IgE)
> 0.35 U/mL], maternal smoking during pregnancy,
and presence of older siblings in the household.
Additional
variables considered for inclusion in the
multivariate analysis included report of any of
the following in the child’s first year of life:
any physician-diagnosed lower respiratory tract
infections (pneumonia, croup, bronchitis, or
bronchiolitis), as determined by bimonthly
questions to the child’s primary caregiver
(Celedon et al. 1999); visible water damage;
mold or mildew inside the home; and report of
water damage in the basement, presence of
concrete floors in the baby’s room, use of a
dehumidifier, and use of an air conditioner. A
composite variable representing the presence of
either water damage or visible mold or mildew in
the home in the child’s first year of life was
also examined.
Statistical methods. All final models
reported used survival analysis (Cox
proportional hazards regression) to explore time
to first report of doctor-diagnosed allergic
rhinitis or hay fever. We performed initial
exploratory analyses of univariate associations
between allergic rhinitis and potential
predictor variables using logistic regression,
chi-square analyses, and Cox regression. We
performed all statistical analyses using SAS
statistical software (version 8.2, SAS Institute
Inc., Cary, NC) and S-Plus (version 6.1,
Insightful Corporation, Seattle, WA).
Multivariate models were constructed that
included significant (p≤ 0.05) fungal
taxa, significant independent predictors of
allergic rhinitis, and variables that may
confound the relationship between the fungal
levels and allergic rhinitis. The proportional
hazards assumption was validated in final
models.
Lasso
approach. Multivariate models were
independently validated using the lasso approach
for model shrinkage described by Tibshirani
(1997). This technique prevents the tendency to
overfit a model by constraining the effect that
the variables will have in the model. This is
one approach used to combat the
multiple-comparison hazards that arise when so
many different taxa of fungi need to be
considered.
Of the 405
children included in this analysis, 52 (12.8%)
were diagnosed with allergic rhinitis or hay
fever by a doctor before or at 5 years of age.
Table 1
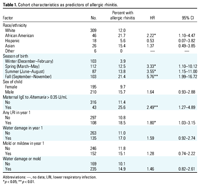 |
Table 2
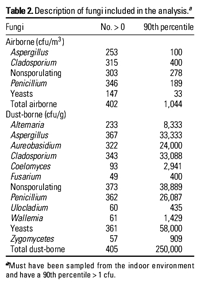 |
Table 3
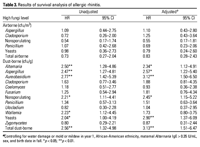 |
Table 4
 |
Relationship
between cohort characteristics and allergic
rhinitis. Table 1 shows the relationship
between cohort characteristics and
doctor-diagnosed allergic rhinitis by 5 years of
age. Factors associated with an increased risk
of allergic rhinitis included African-American
ethnicity, being born between September and
November, maternal sensitization to
Alternaria (IgE to Alternaria > 0.35
U/mL), and having at least one lower respiratory
tract illness in the first year of life. Male
sex and water damage or mold or mildew in the
building of residence within the first year of
life were marginally associated with
doctor-diagnosed allergic rhinitis by 5 years of
age.
In Table 2, we
report the number of houses from which each
taxon was cultured, and the value corresponding
to the 90th percentile. Penicillium was
the most common taxon sampled from the air, but
Cladosporium, Penicillium, and
nonsporulating fungi had the highest levels
recovered from the air. From the dust samples,
Aspergillus was most commonly recovered
sporulating taxon, followed by Cladosporium.
High levels of
dust-borne Aureobasidium, Aspergillus,
Alternaria, yeasts, and nonsporulating
fungi as well as high levels of total fungi were
independently associated with the development of
doctor-diagnosed allergic rhinitis by 5 years of
age in analyses controlling for factors
significantly associated with allergic rhinitis
(Table 3). These factors included water damage
or mold or mildew in the building during the
first year of the child’s life, any lower
respiratory tract illness in the first year,
male sex, African-American race, birth date
between September and November, and maternal IgE
to Alternaria > 0.35 U/mL.
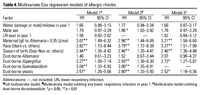
In a
multivariate adjusted proportional hazards
regression analysis looking simultaneously at
the effects of high fungal levels and
significant independent predictors (Table 4),
dust-borne Aspergillus [hazard ratio (HR)
= 3.27; 95% confidence interval (CI),
1.50-7.14], Aureobasidium (HR = 3.04; 95%
CI, 1.33-6.93), and yeasts (HR = 2.67; 95% CI,
1.26-5.66) remained significantly associated
with allergic rhinitis. Alternaria and
Cladosporium were not significant in the
multivariate model, because they are highly
correlated with other fungi that were
significant. Alternaria was significantly
correlated with Aureobasidium (R =
0.33, p < 0.0001) to the point where its
contribution to the model is not statistically
significant after considering the effect of
Aureobasidium. When Aureobasidium is
omitted from the model (Table 4, model 3),
Alternaria is borderline significant (HR =
2.07; 95% CI, 0.98-4.37). Alternaria and
Cladosporium were highly correlated as
well (R = 0.30, p < 0.0001).
Having high levels of total dust-borne fungi was
not significant in multivariate models that had
the specific taxa listed above (p =
0.44). The magnitude and strength of these
associations remained essentially unchanged when
controlling for the presence of dust mite,
cockroach, and cat allergens and endotoxin in
the home, none of which had significant
independent associations with allergic rhinitis
by age 5 in multivariate models (data not
shown).
High dust-borne
fungal levels of Alternaria,
Aspergillus, Aureobasidium, and
yeasts measured in the home in the first 3
months of life were associated with the
subsequent development of doctor-diagnosed
allergic rhinitis within the first 5 years of
life, independent of water damage or mold or
mildew in the home, race, sex, season of birth,
lower respiratory tract infection in the first
year of life, and maternal IgE to Alternaria
> 0.35 U/mL. Home dampness has been shown to be
related to allergic rhinitis exacerbations
(Kilpelainen et al. 2001), but to our knowledge,
the relation of either water damage or
mold/mildew or high levels of specific fungal
taxa to the development of allergic rhinitis in
childhood has not previously been reported.
Although other
studies have found associations between measures
of total fungi and health outcomes (Gent et al.
2002), it is likely that, in our study, “total
fungi” is a noisier estimate of relevant fungal
exposures than are our other metrics of fungi.
The measure of “total fungi” would combine fungi
not likely to be related to rhinitis along with
fungi that have been identified as being related
to allergic sensitization. This could lead to
less precise estimates of fungal heath effects
and an attenuation of the relationship between
fungi and allergic rhinitis.
Fungal exposure
is complex. Fungi can contain allergens,
irritants, toxins, and sometimes even
potentially infectious units (Johnston 2000). (1 3)-β-d-Glucans--glucose
polymers that are structural components of most
fungal cell walls--are known to stimulate
macrophages and neutrophils and have been shown
to be good markers for the overall levels of
fungal concentrations in floor dust (Chew et al.
2001). Beijer et al. (2002) found that an
inhalation challenge to (1
3)-β-d-Glucans--glucose
polymers that are structural components of most
fungal cell walls--are known to stimulate
macrophages and neutrophils and have been shown
to be good markers for the overall levels of
fungal concentrations in floor dust (Chew et al.
2001). Beijer et al. (2002) found that an
inhalation challenge to (1 3)-β-d-glucans
has an effect on the inflammatory cells, which
may be related to chronic exposure to household
molds. Fungi are ubiquitous, and exposure is
impossible to completely avoid. Often, there are
no adverse effects from these exposures (Macher
2000), but at times exposure to fungi can
directly and indirectly influence an
individual’s health. Fungal exposure in
sensitized individuals may result in histamine
and IgE-mediated nasal inflammation (Skoner
2001). Chronic exposure to fungi may also cause
chronic nasal symptoms that are not allergic in
nature, but rather caused by irritants (Daisey
et al. 2003; Dotterud et al. 1996). Symptoms of
allergic rhinitis are similar to the chronic
nasal symptoms often found with sick building
syndrome. Given that fungi may play a role in
sick building syndrome as well (Scheel et al.
2001), differentiation may be difficult.
3)-β-d-glucans
has an effect on the inflammatory cells, which
may be related to chronic exposure to household
molds. Fungi are ubiquitous, and exposure is
impossible to completely avoid. Often, there are
no adverse effects from these exposures (Macher
2000), but at times exposure to fungi can
directly and indirectly influence an
individual’s health. Fungal exposure in
sensitized individuals may result in histamine
and IgE-mediated nasal inflammation (Skoner
2001). Chronic exposure to fungi may also cause
chronic nasal symptoms that are not allergic in
nature, but rather caused by irritants (Daisey
et al. 2003; Dotterud et al. 1996). Symptoms of
allergic rhinitis are similar to the chronic
nasal symptoms often found with sick building
syndrome. Given that fungi may play a role in
sick building syndrome as well (Scheel et al.
2001), differentiation may be difficult.
In previous
articles we have reported that endotoxin was a
risk factor for wheeze (Park et al. 2001) yet
had a protective effect on eczema in the first
year of life (Phipatanakul et al. 2004).
However, the associations of fungi with allergic
rhinitis remained essentially unchanged when
controlling for the presence of indoor allergens
and endotoxin.
There is a
large literature demonstrating the relationship
between fungal sensitization and allergic
rhinitis and asthma, as well as a growing
literature linking fungal exposure to asthma and
rhinitis exacerbation (Downs et al. 2001;
Halonen et al. 1997) in sensitized children
(Arshad et al. 2001; Nolles et al. 2001).
However, the fact that children with allergic
rhinitis are sensitized to fungi does not
necessarily mean that fungal exposure will lead
to allergy or allergic symptoms. This study
suggests a prospective relationship between
exposure and symptoms, although elevation of the
individual fungal taxa may not lead directly to
symptom development. For example, relative
elevation of specific taxa such as
Aspergillus, Aureobasidium, and
yeasts may be a marker for overall fungal
exposure. In models containing the individual
taxa and our measure of total fungi, total fungi
was not predictive of allergic rhinitis,
suggesting that the combination of individual
taxa was a better marker of clinically relevant
fungal exposure.
Sensitization
to Alternaria, Aspergillus, and
Penicillium allergens have been suggested as
being related to allergic rhinitis and asthma
exacerbation in children (Clark et al. 1999;
Halonen et al. 1997). Purified and characterized
allergens have been prepared for Aspergillus
fumigatus (Asp f 1 and Asp f
3) (Macher 2000), which has been shown to cause
skin reactivity in patients with allergic
rhinitis (Johnston 2000). However, A.
fumigatus was sampled from only three
houses. The most commonly sampled species in
this cohort were members of the Aspergillus
glaucus group (n = 275) and
Aspergillus versicolor (n = 255).
Although skin test material is commonly
available for both, less is known about the
allergenicity of these species. A. versicolor has
been implicated in an outbreak of allergic
respiratory disease (Jarvis and Morey 2001).
Cladosporium
and Alternaria are known sources of
allergens, including the proteins Alt a
1, Alt a 2, and Cla h 1 (Johnston
2000). Many cross-sectional studies have
correlated Cladosporium and Alternaria
sensitization with diagnoses of asthma, eczema,
and rhinitis (Arshad et al. 2001; Downs et al.
2001; Halonen et al. 1997; Nolles et al. 2001).
Aureobasidium has been associated with
hypersensitivity pneumonitis (Greinert et al.
2000), but its role in the development of
allergic rhinitis is unknown. In this study, it
may be a marker for mold in the home, rather
than the actual source of the chronic nasal
symptoms.
In their
association with chronic nasal symptoms, yeasts
may also be a marker for overall mold exposure,
because although they have known associations
with skin manifestations of allergy, nasal
symptoms are less commonly documented. Yeasts
are unicellular fungi that reproduce primarily
by budding and, in culture, form pasty colonies
similar to those of bacteria (Macher 2000).
Several studies have shown that patients with
atopic dermatitis and respiratory allergy often
develop IgE antibodies against yeasts (Devos and
Van der Valk 2000; Nittner-Marszalska et al.
2001; Tengvall Linder et al. 2000). One showed
that patients with atopic dermatitis of the head
and neck developed IgE antibodies against the
yeast Pityrosporum ovale much more often
than did their control group. Another found that
the enzyme enolase from baker’s yeast (Saccharomyces
cerevisiae) induces an immediate dermal
allergic reaction in subjects with respiratory
allergy and positive skin tests to Candida
albicans and other fungi. A third group
demonstrated that Pityrosporum orbiculare can
induce an eczematous reaction in sensitized
atopic dermatitis patients.
Potential
study limitations. This study does have
some limitations. Potential limitations of our
work include the relatively small sample size,
along with the multiplicity of variables being
considered. Our use of the Lasso approach
provides an efficient and appealing method for
handling multiplicity issues. The Lasso method
is much less prone to missing real associations
than more traditional adjustments for multiple
comparisons, such as Bonferroni. Even so, the
reality is that with only 52 cases, some
important differences may have been missed. It
is unlikely, however, that our analysis
erroneously identified many false associations,
if any. In addition, because of the collinearity
of the fungal data and the paucity of events, it
is difficult to assess the risk of each taxon
controlling for the others. For example,
Alternaria is so significantly correlated
with Aureobasidium that its effect is
masked when both are included in the model. Even
still, in a combined multivariate model, high
levels of Aspergillus, Aureobasidium,
and yeasts were independently associated with an
increased risk of doctor-diagnosed allergic
rhinitis within the first 5 years of life. In
addition, the air and dust samples were single,
short-term samples. Therefore, it is not clear
how representative the samples are of long-term
household fungal levels. We used only a
culture-based analysis to determine fungal
concentrations, which would underestimate actual
fungal spore exposures and does not account for
other fungal agents. Also, the culture media
used for both the air and dust samples, DG18,
has high solute concentration, which limits the
amount of available water. Thus, some strongly
hydrophilic fungi (e.g., Stachybotrys
sp., some yeasts) might be underrepresented. In
addition, all relationships discussed in this
article are related to exposures that occurred
in the first home in which the child lived, and
do not account for conditions in any subsequent
homes or locations where the child may have
spent significant time (e.g., a child care
center). Moreover, without sampling performed
throughout the study (Chew et al. 2001), it is
impossible to determine whether high fungal
levels early in life increase the likelihood of
having doctor-diagnosed allergic rhinitis later
in life, or whether the early sampling is
representative of the environment later in life.
In addition, the absence of definitive
measurements of allergy, and the relatively
early age of ascertainment of outcome in this
study such that some children may yet develop
allergic rhinitis in this cohort, may be
responsible for the lack of an endotoxin effect
on the risk of allergic rhinitis in this
analysis. Further, parental report of allergic
rhinitis or hay fever was not confirmed by chart
review, but primary care physician documentation
of these diagnoses may not be the gold standard
because it is likely to vary by individual
practice. In addition, it is possible that the
parents did not understand or recall a diagnosis
of allergic rhinitis. Lastly, without skin-prick
testing, the possibility exists that nasal
symptoms related to chronic irritation were
misdiagnosed as allergic rhinitis by the
physicians of some of the participating
children. Even if fungal exposure in infancy
leads to allergic rhinitis in some children and
to chronic irritant nonallergic rhinitis in
others, these findings still have important
public health implications.
Recently, more
attention has been given to the need for
monitoring of fungal levels and active
intervention, where necessary (Johnston 2000).
Epidemiologic studies have found associations
between report of home dampness and respiratory
symptoms in early life, but most have not
measured fungi directly and have not determined
whether fungal effects can be distinguished from
other exposures related to dampness that
prospectively predict respiratory disease.
Independent of early life report of home
dampness, we found that specific dust-borne
fungi measurable in the home in the first 3
months of a child’s life was associated with an
increased risk of developing doctor-diagnosed
allergic rhinitis by 5 years of age.



